NHS Vaccine Rollout Success
How spirit, speed and skills conquered some of the biggest challenges
Mark Payne – Health & Social Care Managing Consultant, Practicus
Behind the success of the life-saving vaccine roll-out lies a story of exceptional collaboration and commitment by people across many disciplines, who pulled together at a critical time to take on the challenge of the COVID19 pandemic. From lab to vaccine centre, everyone rallied to get millions of people vaccinated in a matter of months. A historic achievement, something to be proud of and with many complex challenges that had to be overcome.
Roll-Out Challenges ironed out by Experts
Rolling out a vaccination programme to an entire nation is massively challenging, especially when racing against time in order to save lives. Getting enough vaccines off the production lines was only the start of the battle. Getting vaccines to those most at risk and then the wider population needed meticulous organisation. Every minute detail from production, purchase, storage, transportation, equipment as well as finding physical locations and staff to man them, often supported by volunteers needed to be carefully planned with joined up thinking. Added to this was the challenge of utilising technology and data, to monitor and manage flows to prioritise the geographical distribution of often limited stock. The success of the programme was not down to a single solution or person but to many interdependent factors coming together at the right time in the right place – no easy task.
Planning and preparation were crucial and the team at Practicus, are proud to have had the opportunity to play a small, but important role in supporting the NHS at a critical time at the very beginning and throughout the national NHS vaccine roll-out programme. Our highly skilled and experienced Interim Consultants provided much needed supply chain experience, engagement and programme leadership from start to finish. Bringing external and non NHS experience they drew upon knowledge gained from working internationally to deliver complex programmes. This was particularly evident in the delivery of key aspects of vaccine distribution to a diverse range of centres and the swift scaling up of the operation as more vaccine became available to the general public
The magnitude and intensity of such a supply chain challenge should not be underestimated. Early on, NHSE / I understood the importance of using data insights to drive decision-making processes and help shape the strategy and execution of the vaccine rollout programme. This investment in a data store and platform helped to identify how COVID19 spreads so in turn could direct health and care resources to hot spots and optimise the supply of vaccines and equipment in an equitable way.
UK Head Start
The UK Government was quick off the mark with both investment and sufficient procurement of the UK’s vaccine supplies. The early commitment and risk of early investment paid off with the MHRA being the first regulator in the world to approve BioNTech/Pfizer vaccine in December 2020, with the world’s first COVID19 inoculation a week later. This was the culmination of year-long planning by the NHS and launched the mass vaccine rollout, starting with Pfizer. Astra Zenica came onstream in January, Moderna arrived in the UK in April, and delivery of Janssen is expected later in the year.
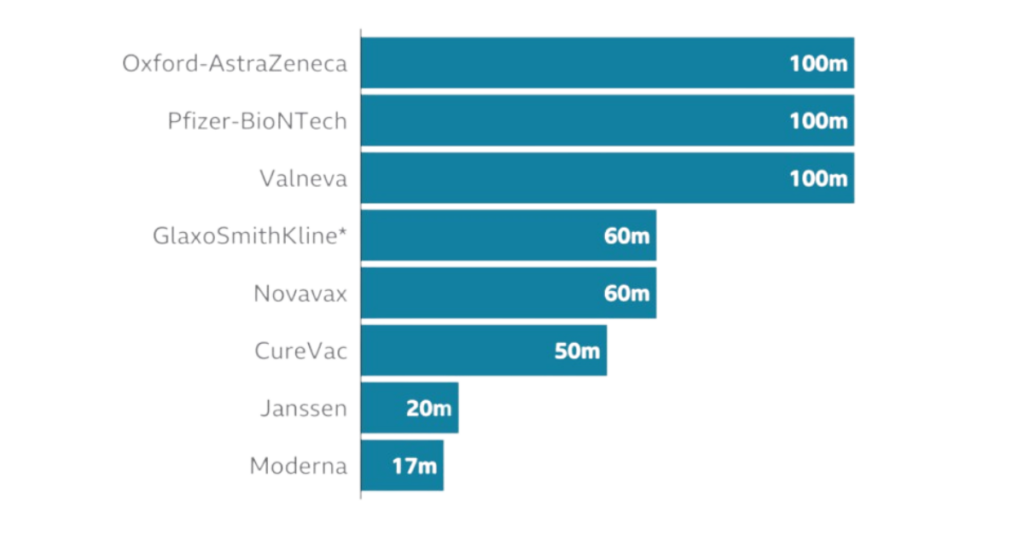
To date, the UK has ordered more than 500 million doses of seven of the most promising vaccines, including the four so far approved for use (Pfizer, AZ, Moderna, Janssen). This includes some reserved for a vaccination booster programme in the autumn. The Government has also invested in manufacturing capacity in the UK. The UK COVID Dashboard keeps track of vaccine take up.
The vaccine roll-out has been highly successful with high levels of take up, however, two key challenges remain:
- Addressing vaccine hesitancy and lower take-up in certain groups (ethnic minorities and young)
- Keeping ahead of potential variants while large parts of the globe still need to get vaccinated.
The NHS is working hard to manage the first, in the knowledge that the current vaccine version is not failproof, and boosters are likely to be needed. The second, will remain a reality as long as unvaccinated parts of the world remain.
Vaccine Hubs
Early investment and clever procurement alone do not account for the UK’s vaccine rollout triumph, it could be argued that the combined strengths of NHS’s national planning capabilities, local infrastructure and passion shown at every level are key differentiators. The ability to reach out and use GP surgeries, pharmacy sites, hospital hubs, whilst turning large venues into mass vaccination sites was critical in addressing reach at speed. In addition, pop-up sites and mobile healthcare professionals delivered the vaccine to hard-to-reach patients in places such as care homes and prisons.
To ensure success, NHS leaders brought in both the military and private sector support to plug key logistic skills and project management gaps. Clearly the commitment and ability to get the right healthcare staff in the right place with the right equipment and the patients to vaccinate in the same place at the same time, is a multi-layered challenge.
Strategically located vaccination hubs have been pivotal in promoting vaccine uptake and this was an important consideration in location sourcing. Once selected, each site needed to be transformed into an efficient centre and supplied with the right equipment and personnel. Even before receiving vaccines, each centre needed to be fitted out and consistently supported to deliver a unified vaccine programme. This is one of the areas where the NHS England CV19 data store made a real difference by keeping track of areas such as warehouse inventories and staff readiness.
A national distribution service has been set up to ensure that every vaccination site receives supplies of vaccines, consumables and PPE that they need.
The number of centres ramped up quickly to include stadiums, community halls, even places of worship – in fact anywhere where vaccines could be delivered to meet local population needs. As many as 30,000 NHS workers and 100,000 volunteers stepped forward to vaccinate the population. In July 2021, there were over 2,000 sites where people could get vaccinated. While some larger sites were being decommissioned to return to their original purpose, many more pop-up and walk-in centres are constantly being created, bringing the vaccine to the general population. The logistical challenge for the planners to ensure just the right number of vaccines are available should not be underestimated – and reduction or total elimination of wasted vaccine made a priority.
Logistics
The speed from creating a vaccine in the lab to manufacturing and distribution was unprecedented, but the race from manufacturer to vaccine centre is arguably even more time sensitive and complex.
Key considerations at the heart of vaccine supply chain management and roll out include:
- Inventory Management
Securing delivery of contracted quantities to agreed timescales underpins the whole immunisation programme. Any production issues needed to be flagged promptly and reflected in the supply chain plans. An effective inventory management system is a vital tool to keep track of vaccine stock and other consumables and PPE, both in depots and at vaccine centres. - Transportation
The cold storage requirements of the Pfizer and Moderna vaccines made distribution more challenging. The Pfizer vaccine needs to be stored at a temperature of at least -70 °C. Fortunately, Pfizer has designed special cases filled with dry ice which hold between 1,000 and 5,000 doses at the right temperature, so they can be transported from their production facility in Belgium without the need for refrigerated trucks. But something as simple as a shortage of dry ice could have had a major impact (not least as every country was also facing the same challenges). With a short shelf life of just 10 days accurate planning and distribution was of critical importance. - Dosing Dilemma
Unlike the 15m Flu vaccines that the NHS is used to dispensing every year, all the COVID19 vaccines currently being used in the UK, require two doses, adding to the logistics challenge. The first dose primes the immune system and the second dose (usually administered a few weeks after the first) boosts it. Getting the right number of doses to sites to meet the needs of those invited is crucial. Planners needed to be able to quickly react to key policy changes such as moving vaccine interval times. Another significant change was the decision not to give Astra Zenica to the cohorts below first 30, then 40 years of age. All these changes needed to be quickly filtered into the scheduling of vaccines to the vaccine centres.
Keeping track of who got vaccinated, when and which vaccines (as both doses currently need to come from the same company), and when the next one is due is another daunting logistical challenge. This is where having data platforms in support saves the day. - Booking System
Existing GP patient records/databases were used to invite patients to book as their age or needs profile became eligible. Online booking systems were put in place for vaccine centres is a complex area in its own right with the need to allow cancellations and re-bookings at short notice. Matching vaccine supply with availability at centres was no mean feat. More recently innovations such as pop-up walk-in centres without the need to book are being quickly erected and hard to reach population groups tackled.
- Vaccine Preparation on Site
The Pfizer vaccine does not arrive with pre-filled syringes, unlike the annual flu vaccine, so trained health-workers are needed to dispense it from vials to then use. Once defrosted, the doses can be held in a standard refrigerator, at between 2C and 8C but possibly for just 24 hours. These vital needs and timing sensitivities have had to be tightly factored into the project plans. - Avoiding Wastage
With vaccines being so scarce, avoiding vaccine wastage is key. Using low wastage syringes helped to get 10% more doses out of the vials than projected. Staff training and empowerment minimises precious vaccines going to waste. Furthermore, stock availability has to be coordinated with the processing capacity. To ensure all stock is used up before it deteriorates, contingency measures like overbooking appointments, or patient availability at short notice need to be in place. Our team is particularly proud of their achievement in optimising the supply chain and mitigating the potential for wastage by proactively diverting stock as required as well as supporting infrastructure and PPE. - Volunteers
An army of willing helpers have played a very important role throughout. Two portals, one for healthcare workers and one for willing helpers offering support in the mass vaccination hubs were created to co-ordinate availability to help give vaccines, or manage patient flow. A similar system supported community-based volunteers for example with medicine deliveries. - Technology
Technology has undoubtedly been a key enabler of the success of the program providing more single source of truth data, than in any other Government programme. The analytics platform and purpose-built data store has blown open visibility of the end-to-end supply chain, with particular focus on tracking. This enables vaccination sites to understand their planned delivery times and allows them to schedule time-critical clinics.
Future
The World Health Organisation (WHO) has stated that the COVID19 virus may be something that we continue to live with and thus we may need to instigate a regular vaccine programme to maintain immunity against this virus. Doing so will require the development of systems to manage vaccination levels, connecting with existing patient healthcare records but also connecting with vaccine production, production location holding, transportation scheduling, local storage information etc. Technology is a critical vaccine programme enabler, but it takes skilled project management to make sure that available resources are used and plans executed in the best way.
It is increasingly part of Government policy to roll out a booster programme this autumn initially for those above 50. This will determine the scale of what is needed, the digital solutions needed and the people skills required. Not everyone is eligible for the annual flu jab, so the COVID booster programme will be bigger, and again more logistically complex to deliver.

Mark Payne
Health & Social Care Managing Consultant
LinkedIn
Our unrivaled client and candidate support have made us the first choice for healthcare appointments. Get in touch if you wish to find out more.
